 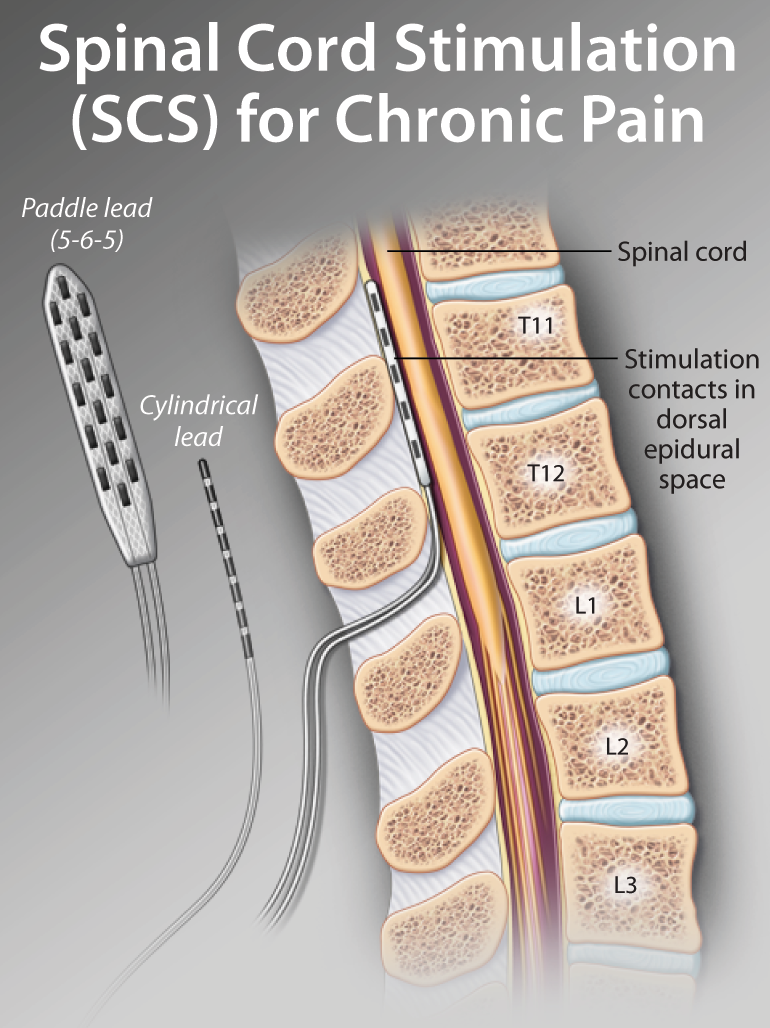
Iatrogenic issues related to technique numbered five events: dural tear during implant, lead damage during implant, anterior migration of lead during implantation, suboptimal connection of the extension to the generator, and a cap not installed on the generator when one lead was implanted. Biological complications totaled 16 events and included infection or wound breakdown, pain at the incision site, and fluid collection in the pocket. Six patients had loss of therapeutic effect, loss of paresthesia, or unpleasant paresthesia. Hardware-related issues, including lead migration, damage to the leads, and generator migration, accounted for 13 of the events. Surgery was required to resolve the issues of 20 (24%) patients. A total of 27 (32%) patients experienced a total of 40 device-related complications. Ultimately 84 patients received an electrode during the 12 months of the study. One hundred patients were randomized to SCS or CMM, and cross over was permitted. They compared conventional medical management (CMM) to SCS for patients with failed back surgery syndrome resulting in residual predominant leg pain. Kumar and associates 9 published a prospective study regarding SCS in 2007. Table 15-2 Device-Related Complications from Three Prospective Randomized Spinal Cord Stimulation Studies 4 According to the systematic review by Turner and associates 5 of 22 studies involving patients with diagnoses, including failed back surgery syndrome or complex regional pain syndrome ( Table 15-1), complication rates associated with SCS are noted to be 34%. In an epidemiological report published in 1999, more than 1 million injuries and nearly 100,000 deaths occur annually as a result of errors in medical care. 3 When appropriate, recommendations will be proffered. Increased awareness of potential contributors of untoward events may help to decrease the likelihood of complications. 
Anecdotal information can also be useful as a way to increase awareness. The goal of this chapter is provide information regarding the types and prevalence of complications and trends noted. Unfortunately complications of SCS occur just as complications occur with all other interventional procedures. 2 The majority of patients have a positive outcome involving a reduction of pain with no untoward events. In 2007 it is estimated that a total of 27,484 SCS implants occurred over all major payer types in the United States. 1 With technological advances, SCS has become an increasingly effective treatment for those with many types of chronic pain conditions. Spinal cord stimulation (SCS) is a treatment for chronic intractable pain that was introduced by Shealy in 1967. Most complications with SCS can be successfully treated or surgically revised when recognized early and given proper attention. Allergic reaction to the device has also been documented. Intravenous antibiotics before surgery can reduce this risk, but infection can arise even months or years after implantation when organisms lurk inside a device. Biological complications generally consist of infection or wound breakdown but can also include pain at the site. Implantations in the cervical spine are apparently more subject to migration because of the increased mobility relative to lumbar implantations. Loss of paresthesia or unpleasant paresthesias can indicate these technical complications. Risks can increase with repeated surgeries. Device-related complications can include lead migration, generator migration, or damage to the leads and generally require surgery to repair or replace the device. One reported mean complication rate is 36%, with complications classified as technical, biological, and other types. This chapter addresses these potential pitfalls in successful SCS implantation, which can arise at any phase of treatment from patient selection to stable use of an implanted device. This minimally invasive surgical procedure is subject to complications, which can be avoided with awareness and vigilance. However, it requires implantation of an electrode array and its associated power source. Chapter Synopsis: Electrical stimulation of the spinal cord (SCS) is generally a safe and effective treatment for neuropathic pain and other conditions. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
